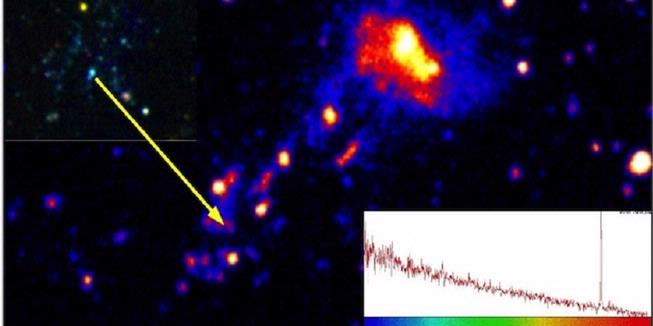
Galaksi IC 3418 yang jatuh ke gugus Virgo. bergerak dengan kecepatan tinggi menyisakan jejak gas dingin
Pada 55 juta tahun lalu, sebuah bintang dilahirkan dalam lingkungan yang sangat liar dan keras. Ia terbentuk dalam jejak panjang yang terbentuk dari gas yang lepas dari galaksi IC 3418. Gas tersebut lepas ketika galaksi IC 3418 melaju cepat menuju gugus Virgo dan berinteraksi dengan plasma panas di sekitar medium antargugus.
Bintang yang terbentuk itu merupakan bintang biru maharaksasa yang berada di rasi bintang Virgo. Bintang yang berada pada jarak 55 tahun cahaya ini sekaligus menjadi bintang terjauh yang berhasil diamati. Tidak mudah memang. Sebab, untuk bisa mengamati bintang maharaksasa biru tersebut, duo astronom Youichi Ohyama (Academia Sinica, Taiwan) dan Ananda Hota (UM-DAE Centre for Excellence in Basic Sciences, India) harus melakukan pengamatan dengan menggunakan beberapa instrumen.

Citra Jupiter dan komet SL9 hasil tangkapan Teleskop Hubble
Hasil penelitian yang dipublikasikan di jurnal Astronomy & Astrophysics mengungkap bahwa atmosfer Jupiter juga memiliki air. Air berasal dari komet Shoemaker-Levy 9 (SL9) yang menumbuk Jupiter 20 tahun lalu.
SL9 ditemukan mengorbit Jupiter oleh astronom bernama David Levy dan Carolyn serta Eugene M Shoemaker pada 24 Maret 1993. Komet ini adalah komet pertama yang ditemukan mengorbit planet, bukan Matahari.
Analisis lebih lanjut kemudian menunjukkan bahwa SL9 pernah lewat sangat dekat dengan Jupiter pada Juli 1992. Saat itulah, planet terdampak gaya tidal Jupiter hingga terpecah menjadi 21 bagian.
Komet selanjutnya juga lewat dekat Jupiter lagi pada tahun 1994. Saat itu, komet menumbuk Jupiter di wilayah dengan koordinat 44 derajat Lintang Selatan Jupiter. Tumbukan menimbulkan fenomena layaknya bola api.
Penelitian ilmuwan sejak puluhan tahun lalu menunjukkan adanya uap air di Jupiter. Namun, saat itu ilmuwan belum bisa memastikan apakah memang air berasal dari SL 9 atau dari cincin planet ataupun bulan-bulan milik Jupiter.
Studi terbaru yang dilakukan oleh T Cavalie dan rekannya, seperti diberitakan Science Daily, Selasa (23/4/2013), memastikan bahwa air di atmosfer Jupiter memang berasal dari SL9. Cavalie melakukan observasi dengan Herschel Space Observatory milik European Space Agency (ESA).
Dalam publikasinya, Cavalie menguraikan bahwa air tidak tersebar merata di Jupiter. Lebih banyak air terdapat di bagian selatan Jupiter. Cavalie mengungkapkan, 95 persen air yang terobservasi saat ini di Jupiter berasal dari komet SL9.
Lalu, apakah dengan adanya air di atmosfer itu Jupiter bisa mendukung kehidupan? Jupiter adalah planet yang terlalu jauh dari Matahari. Selain itu, Jupiter juga merupakan planet gas. Jadi, jangan berharap Jupiter bisa mendukung kehidupan dalam persepsi manusia saat ini.
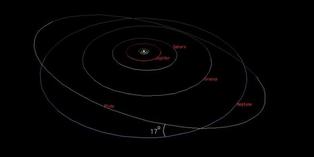
Pluto, the celestial bodies are now classified as a dwarf planet, has many unique. One of the uniqueness is in its orbit around the Sun as the center of the Solar System.
Can be seen in the picture above, Pluto has a different orbits of the planets in the Solar System. Pluto's orbit "steep", have a difference angle 17 degrees. And, it can be seen that there is the point of intersection between the orbits of Pluto and Neptune.
Texas.If you buff Harry Potter novels and movies, you might already be familiar with the cloak that could make Harry Potter "disappeared". Now, the cloak is not just a fantasy. A team of researchers at the University of Texas at Austin created the robe.
Andrea Lu creation cloak could make objects invisible despite only being able to be applied in light of microwaves.

MOSCOW: The number of fighters of the fifth generation Sukhoi T-50 PAK will be increased to 14 units from now on only three. Russia decided to increase the number of the most technologically advanced aircraft in the world to test fly deep.
Russian Air Force Commander Alexander Zelin on Monday, according to RIA Novosti, said, "There have been three fighters who took part in the flight test, the other three expected to be tested in the near future. Entire number of aircraft for flight test 14 units."
Seed plants have flowers. Interest in plant seeds serve as generative reproduction. In the flowers are male and female genitals. Male genitals in a flower seed plants consisting of a stamen (stamens). Has stamen anthers (anthera) that is located at the end of stalk juice (filament). In anthers with one or more space juice (puzzle) which is the formation of pollen (pollen). Pollen is called the male gamete. Genitals are female (pistil) can be composed of ovary (ovarian) stalk buds (stillus), and head Pistil (stigma). Will the fruit is on the plant seeds as the presence of ovules. Plant ovary lies at the base of flower seeds (reseptakel) and shape bubble.

John Dalton was a British teacher. He put forward the theory of atoms based on the Law of Conservation of Mass (proposed by Antonie Lavoiser) which states that in a reaction, the reaction mass before and after the same are subscription and Comparative Law Fixed (presented by Josepht Proust) which states that in a pure chemical substance, the ratio of the mass of the element elements in each compound are fixed. For example, is made up of 2 parts water and 1 part H O to form H2O or 1 gram H vs 8 grams of O. Where else would we get water composed of elements with the same ratio.
Dalton's atomic theory includes four (4) as follows:
Each chemical element arranged on small particles that can not be broken again called atoms. Atoms can not be created nor destroyed during a chemical change. Atoms are round like a ball.
Constituent atoms of an element are identical in mass (weight) and certain properties. However, atoms of one element differ from the atoms of other elements.
When forming compounds, different elements joined by a simple comparison. For example, an atom A with an atom B (AB), or one atom A to atom B two (AB2), and so on.
A chemical reaction is merely a displacement of atoms from one set to the other combinations. Atoms themselves individually and always remains unchanged.
Dalton's atomic theory gives details and a more complete explanation than Democritus statement. But this theory is also not perfect. Why is that? The answer please see Atomic Model: JJ Atomic Theory Thomson.
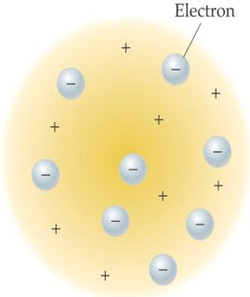
Physicist Joseph John Thomson (1856-1940) was a scientist who was born in Cheetham Hill, where at that place anyway Thomson was named professor of experimental physics since 1884. The study did result in the discovery of the electron Thomson. He knew that the gas is a substance that is able to deliver electricity. Thomson also one of the pioneers of nuclear physics. Thomson won the Nobel Prize for physics in 1906.
This article is a continuation of the previous discussion as well as to answer questions in writing Atomic Model: Atomic Theory Dalton. Dalton's atomic theory long held by scholars at that time until the discovery of negatively charged electrons by JJ Thomson in 1897 (see History of discovery in Proton, Neutron, and Electron). The discovery of electrons finally broke Dalton theory that atoms are the smallest matter. Because the negativity charged electrons Thomson thinks that there is a positive charge as a counterweight. Thus the atom is neutral.
Thomson atomic model illustrates that the atom was a positively charged sphere. Meanwhile, the electrons (the negatively charged atoms) are spread evenly on the surface of the ball. Negative charges are spread out like raisins in raisin bread. The number of positive charges equal to the amount of negative charge so that the atom is neutral.
The number of positive charges = Number of negative charge
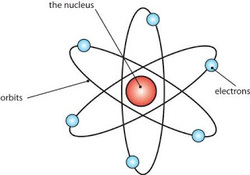
Ernest Rutherford was a chemist who received the Nobel Prize in 1908. He investigated about solving elements and radioactive materials. In 1911, Rutherford with two students (Geiger and Ernest Marsden) has been experimenting with shooting a thin layer of gold using alpha particles, which are known as alpha-ray scattering experiments.
The results of this experiment successfully completed teacher Rutherford atomic theory, namely JJ Thomson. Rutherford model of the atom proposed as follows:
Most of the mass and the whole positive charge contained in an atom is concentrated in a very small region called the nucleus. Atom itself is mostly a blank space.
The amount of positive charge is different from one atom to another atom.
The number of electrons around the nucleus of an atom is like the positive charge on the nucleus. Atom as a whole is neutral.








 RSS Feed
RSS Feed